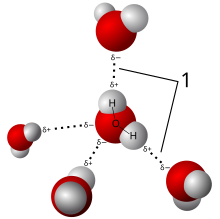
A hydrogen bond is a partially electrostaticattraction between a hydrogen (H) which is bound to a more electronegative atom such as nitrogen (N), oxygen (O), or fluorine (F), and another adjacent atom bearing a lone pair of electrons.
Hydrogen bonds can occur between molecules (intermolecular) or within different parts of a single molecule (intramolecular).[1]Depending on the nature of the donor and acceptor atoms which constitute the bond, their geometry, and environment, the energy of a hydrogen bond can vary between 1 and 40 kcal/mol.[2] This makes them somewhat stronger than a van der Waals interaction, and weaker than fully covalent or ionic bonds. This type of bond can occur in inorganic molecules such as water and in organic molecules like DNA and proteins.
Intermolecular hydrogen bonding is responsible for the high boiling point of water(100 °C) compared to the other group 16 hydrides that have much weaker hydrogen bonds.[3] Intramolecular hydrogen bonding is partly responsible for the secondary and tertiary structures of proteins and nucleic acids. It also plays an important role in the structure of polymers, both synthetic and natural.
In 2011, an IUPAC Task Group recommended a modern evidence-based definition of hydrogen bonding, which was published in the IUPAC journal Pure and Applied Chemistry. This definition specifies:
By Er. Neeraj K.Srivastav (jee mains qualified)

Comments
Post a Comment